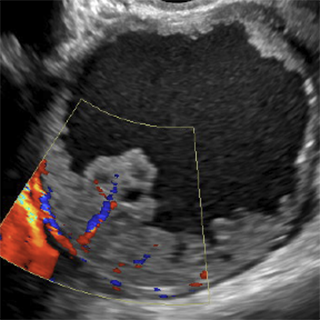
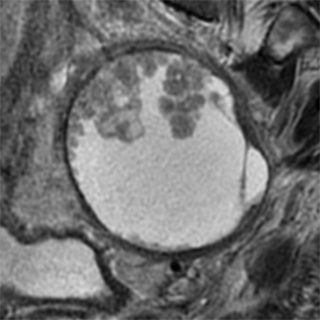
 O-RADS™ is an acronym for Ovarian-Adnexal Imaging-Reporting-Data System, a clinical support system for the standardized description and classification of ovarian/adnexal lesions.
O-RADS™ is an acronym for Ovarian-Adnexal Imaging-Reporting-Data System, a clinical support system for the standardized description and classification of ovarian/adnexal lesions.
The system consists of two complementary arms:
- Ultrasound (US) - the primary imaging modality, which is optimized for high sensitivity
- Magnetic Resonance Imaging (MRI) - a secondary tool that allows higher specificity.
Both O-RADS US and O-RADS MRI employ lexicon terminology for describing the imaging characteristics of ovarian/adnexal lesions and data-based risk assessment categories to assign a risk of malignancy. The goal of the O-RADS system is to optimize ovarian cancer outcomes while minimizing unnecessary surgery in patients with adnexal lesions.